 
The excited state is unstable, and the electron jumps to a lower level while releasing a photon. H : Planck’s constant ( = 6.626 x 10 -34 J.s)īohr was able to apply this quantization idea to his atomic orbital theory and found that the orbital energy of the electron in the n th orbit of a hydrogen atom is given by,Īccording to the Bohr model, electrons can only absorb energy from a photon and move to an excited state if the photon has an energy equal to the energy difference between the two energy levels. The energy carried by a photon is given by, When it exists as a particle, it is called a photon. According to this theory, electromagnetic radiation can exist as both a wave and a particle. Only shells of radii given by the above equation are allowed.īy the early 20 th century, physicists Max Planck and Albert Einstein developed the quantum theory, which states that matter absorbs and emits radiation in discrete packets known as quanta. N : principal quantum number (n = 1,2,3…) R B : smallest electron radius of the hydrogen atom, known as Bohr radius ( = 0.529 x 10 -10 m) The radius of the n-th orbit is given by, The radius, angular momentum, and energy take specific values only. According to Bohr, the orbits in which the electrons reside are quantized. He proposed that when an electron jumps from a higher orbit to a lower orbit or one closer to the nucleus, it emits radiation in the form of light.īohr resolved the fallacy of the classical model by incorporating Planck’s quantum theory and Einstein’s photoelectric effect. While observing the emission spectra of the hydrogen atom, Bohr was able to account for the fine spectral lines. According to Bohr, electrons orbit around the nucleus in discrete circular orbits with well-defined radii, which depend on the principal quantum number n. He used quantum mechanics to explain the spectral lines emitted by the hydrogen atom. 
Moreover, the classical model considers that electrons revolving around orbits emit continuous radiation, which fails to explain the atomic line spectra of many atoms.īohr applied his ideas to the Rutherford model, and hence, his model is also called the Bohr-Rutherford model. In other words, it does not explain why atoms are stable. 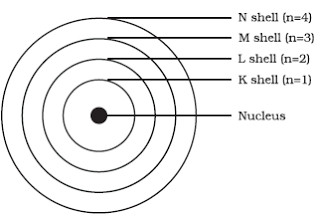
It fails to explain why the electrons do not spiral inwards during their motion and collapse into the nucleus. Rutherford developed his model in 1911, which is also known as the classical model. Several experiments done by New Zealand physicist Ernest Rutherford established that an atom consists of a positively charged nucleus surrounded by a cloud of negatively charged electrons. According to Planck’s relation, when an electron moves from one orbit to another, it absorbs or emits radiation.ĭevelopment of the Bohr Model How is Bohr’s Atomic Model Different from Rutherford’s Model.The energy of the electron depends on the principal quantum number n.Electrons orbiting in orbits do not interact with each other.Electrons move around the nucleus in orbits of well-defined radius.Bohr Model Salient Features of the Bohr Model 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
